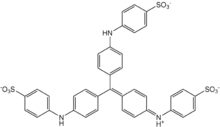
Methyl blue
 | |||
 | |||
| |||
| Names | |||
|---|---|---|---|
| Other names Cotton blue, Helvetia blue, Acid blue 93, C.I. 42780 | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
ChemSpider |
| ||
ECHA InfoCard | 100.044.852 | ||
PubChem CID |
| ||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula | C37H27N3Na2O9S3 | ||
Molar mass | 799.814 g/mol | ||
| Appearance | red solid | ||
Solubility in water | Soluble in water, slightly soluble in ethanol | ||
| Hazards | |||
NFPA 704 |  1 1 0 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
Infobox references | |||
Methyl blue is a chemical compound with the molecular formula C37H27N3Na2O9S3. It is used as a stain in histology,[1] and stains collagen blue in tissue sections. It can be used in some differential staining techniques such as Mallory's connective tissue stain and Gömöri trichrome stain, and can be used to mediate electron transfer in microbial fuel cells. Fungal cell walls are also stained by methyl blue.
Methyl blue is also available in mixture with water blue, under name Aniline Blue WS, Aniline blue, China blue, or Soluble blue.
See also
- Potassium ferrocyanide
- Potassium ferricyanide
- Methylene blue
- Egyptian Blue
- Han Purple
- Gentian violet
- Fluorescein
References
^ R. W. Sabnis (2010). Handbook of Biological Dyes and Stains: Synthesis and Industrial Applications. John Wiley & Sons. p. 24..mw-parser-output cite.citation{font-style:inherit}.mw-parser-output q{quotes:"""""""'""'"}.mw-parser-output code.cs1-code{color:inherit;background:inherit;border:inherit;padding:inherit}.mw-parser-output .cs1-lock-free a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/6/65/Lock-green.svg/9px-Lock-green.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-limited a,.mw-parser-output .cs1-lock-registration a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/d/d6/Lock-gray-alt-2.svg/9px-Lock-gray-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-subscription a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/a/aa/Lock-red-alt-2.svg/9px-Lock-red-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration{color:#555}.mw-parser-output .cs1-subscription span,.mw-parser-output .cs1-registration span{border-bottom:1px dotted;cursor:help}.mw-parser-output .cs1-hidden-error{display:none;font-size:100%}.mw-parser-output .cs1-visible-error{font-size:100%}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration,.mw-parser-output .cs1-format{font-size:95%}.mw-parser-output .cs1-kern-left,.mw-parser-output .cs1-kern-wl-left{padding-left:0.2em}.mw-parser-output .cs1-kern-right,.mw-parser-output .cs1-kern-wl-right{padding-right:0.2em}

