Rheumatoid arthritis
| Rheumatoid arthritis | |
|---|---|
 | |
| A hand affected by rheumatoid arthritis | |
| Specialty | Rheumatology |
| Symptoms | Warm, swollen, painful joints[1] |
| Complications | Low red blood cells, inflammation around the lungs, inflammation around the heart[1] |
| Usual onset | Middle age[1] |
| Duration | Lifelong[1] |
| Causes | Unknown[1] |
| Diagnostic method | Based on symptoms, medical imaging, blood tests[1][2] |
| Differential diagnosis | Systemic lupus erythematosus, psoriatic arthritis, fibromyalgia[2] |
| Medication | Pain medications, steroids, Nonsteroidal anti-inflammatory drugs, disease-modifying antirheumatic drugs[1] |
| Frequency | 0.5–1% (adults in developed world)[3] |
| Deaths | 30,000 (2015)[4] |
Rheumatoid arthritis (RA) is a long-term autoimmune disorder that primarily affects joints.[1] It typically results in warm, swollen, and painful joints.[1] Pain and stiffness often worsen following rest.[1] Most commonly, the wrist and hands are involved, with the same joints typically involved on both sides of the body.[1] The disease may also affect other parts of the body.[1] This may result in a low red blood cell count, inflammation around the lungs, and inflammation around the heart.[1] Fever and low energy may also be present.[1] Often, symptoms come on gradually over weeks to months.[2]
While the cause of rheumatoid arthritis is not clear, it is believed to involve a combination of genetic and environmental factors.[1] The underlying mechanism involves the body's immune system attacking the joints.[1] This results in inflammation and thickening of the joint capsule.[1] It also affects the underlying bone and cartilage.[1] The diagnosis is made mostly on the basis of a person's signs and symptoms.[2]X-rays and laboratory testing may support a diagnosis or exclude other diseases with similar symptoms.[1] Other diseases that may present similarly include systemic lupus erythematosus, psoriatic arthritis, and fibromyalgia among others.[2]
The goals of treatment are to reduce pain, decrease inflammation, and improve a person's overall functioning.[5] This may be helped by balancing rest and exercise, the use of splints and braces, or the use of assistive devices.[1]Pain medications, steroids, and NSAIDs are frequently used to help with symptoms.[1]Disease-modifying antirheumatic drugs (DMARDs), such as hydroxychloroquine and methotrexate, may be used to try to slow the progression of disease.[1] Biological DMARDs may be used when disease does not respond to other treatments.[6] However, they may have a greater rate of adverse effects.[7] Surgery to repair, replace, or fuse joints may help in certain situations.[1] Most alternative medicine treatments are not supported by evidence.[8][9]
RA affects about 24.5 million people as of 2015.[10] This is between 0.5 and 1% of adults in the developed world with 5 and 50 per 100,000 people newly developing the condition each year.[3] Onset is most frequent during middle age and women are affected 2.5 times as frequently as men.[1] In 2013, it resulted in 38,000 deaths up from 28,000 deaths in 1990.[11] The first recognized description of RA was made in 1800 by Dr. Augustin Jacob Landré-Beauvais (1772–1840) of Paris.[12] The term rheumatoid arthritis is based on the Greek for watery and inflamed joints.[13]
Contents
1 Signs and symptoms
1.1 Joints
1.2 Skin
1.3 Lungs
1.4 Heart and blood vessels
1.5 Blood
1.6 Other
2 Risk factors
2.1 Genetic
2.2 Environmental
2.3 Negative findings
3 Pathophysiology
3.1 Non-specific inflammation
3.2 Amplification in the synovium
3.3 Chronic inflammation
4 Diagnosis
4.1 Imaging
4.2 Blood tests
4.3 Classification Criteria
4.4 Differential diagnoses
4.5 Monitoring progression
5 Prevention
6 Management
6.1 Lifestyle
6.2 Disease modifying agents
6.3 Anti-inflammatory and analgesic agents
6.4 Surgery
6.5 Alternative medicine
6.5.1 Dietary supplements
6.6 Pregnancy
6.7 Vaccinations
7 Prognosis
7.1 Prognostic factors
7.2 Mortality
8 Epidemiology
9 History
9.1 Etymology
10 Research
11 References
12 External links
Signs and symptoms
RA primarily affects joints, but it also affects other organs in more than 15–25% of cases.[14] Associated problems include cardiovascular disease, osteoporosis, interstitial lung disease, infection, cancer, feeling tired, depression, mental difficulties, and trouble working.[15]
Joints
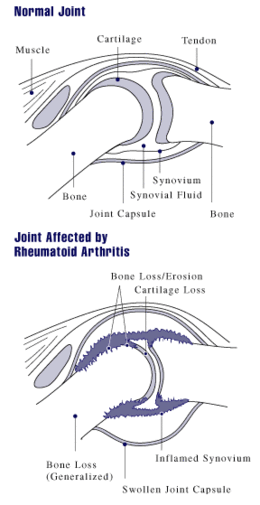
A diagram showing how rheumatoid arthritis affects a joint
Arthritis of joints involves inflammation of the synovial membrane. Joints become swollen, tender and warm, and stiffness limits their movement. With time, multiple joints are affected (polyarthritis). Most commonly involved are the small joints of the hands, feet and cervical spine, but larger joints like the shoulder and knee can also be involved.[16]:1098 Synovitis can lead to tethering of tissue with loss of movement and erosion of the joint surface causing deformity and loss of function.[2]
RA typically manifests with signs of inflammation, with the affected joints being swollen, warm, painful and stiff, particularly early in the morning on waking or following prolonged inactivity. Increased stiffness early in the morning is often a prominent feature of the disease and typically lasts for more than an hour. Gentle movements may relieve symptoms in early stages of the disease. These signs help distinguish rheumatoid from non-inflammatory problems of the joints, such as osteoarthritis. In arthritis of non-inflammatory causes, signs of inflammation and early morning stiffness are less prominent.[citation needed]
The pain associated with RA is induced at the site of inflammation and classified as nociceptive as opposed to neuropathic.[17] The joints are often affected in a fairly symmetrical fashion, although this is not specific, and the initial presentation may be asymmetrical.[16]:1098
As the pathology progresses the inflammatory activity leads to tendon tethering and erosion and destruction of the joint surface, which impairs range of movement and leads to deformity. The fingers may suffer from almost any deformity depending on which joints are most involved. Specific deformities, which also occur in osteoarthritis, include ulnar deviation, boutonniere deformity (also "buttonhole deformity", flexion of proximal interphalangeal joint and extension of distal interphalangeal joint of the hand), swan neck deformity (hyperextension at proximal interphalangeal joint and flexion at distal interphalangeal joint) and "Z-thumb." "Z-thumb" or "Z-deformity" consists of hyperextension of the interphalangeal joint, fixed flexion and subluxation of the metacarpophalangeal joint and gives a "Z" appearance to the thumb.[16]:1098 The hammer toe deformity may be seen. In the worst case, joints are known as arthritis mutilans due to the mutilating nature of the deformities.[18]
Skin
The rheumatoid nodule, which is sometimes in the skin, is the most common non-joint feature and occurs in 30% of people who have RA.[19] It is a type of inflammatory reaction known to pathologists as a "necrotizing granuloma". The initial pathologic process in nodule formation is unknown but may be essentially the same as the synovitis, since similar structural features occur in both. The nodule has a central area of fibrinoid necrosis that may be fissured and which corresponds to the fibrin-rich necrotic material found in and around an affected synovial space. Surrounding the necrosis is a layer of palisading macrophages and fibroblasts, corresponding to the intimal layer in synovium and a cuff of connective tissue containing clusters of lymphocytes and plasma cells, corresponding to the subintimal zone in synovitis. The typical rheumatoid nodule may be a few millimetres to a few centimetres in diameter and is usually found over bony prominences, such as the elbow, the heel, the knuckles, or other areas that sustain repeated mechanical stress. Nodules are associated with a positive RF (rheumatoid factor) titer, ACPA, and severe erosive arthritis. Rarely, these can occur in internal organs or at diverse sites on the body.[citation needed]
Several forms of vasculitis occur in RA, but are mostly seen with long-standing and untreated disease. The most common presentation is due to involvement of small- and medium-sized vessels. Rheumatoid vasculitis can thus commonly present with skin ulceration and vasculitic nerve infarction known as mononeuritis multiplex.[20]
Other, rather rare, skin associated symptoms include pyoderma gangrenosum, Sweet's syndrome, drug reactions, erythema nodosum, lobe panniculitis, atrophy of finger skin, palmar erythema, and skin fragility (often worsened by corticosteroid use).[citation needed]
Diffuse alopecia areata (Diffuse AA) occurs more commonly in people with rheumatoid arthritis.[21] RA is also seen more often in those with relatives who have AA.[21]
Lungs
Lung fibrosis is a recognized complication of rheumatoid arthritis. It is also a rare but well-recognized consequence of therapy (for example with methotrexate and leflunomide). Caplan's syndrome describes lung nodules in individuals with RA and additional exposure to coal dust. Exudative pleural effusions are also associated with RA.[22][23]
Heart and blood vessels
People with RA are more prone to atherosclerosis, and risk of myocardial infarction (heart attack) and stroke is markedly increased.[24][25][26]
Other possible complications that may arise include: pericarditis, endocarditis, left ventricular failure, valvulitis and fibrosis.[27] Many people with RA do not experience the same chest pain that others feel when they have angina or myocardial infarction. To reduce cardiovascular risk, it is crucial to maintain optimal control of the inflammation caused by RA (which may be involved in causing the cardiovascular risk), and to use exercise and medications appropriately to reduce other cardiovascular risk factors such as blood lipids and blood pressure. Doctors who treat people with RA should be sensitive to cardiovascular risk when prescribing anti-inflammatory medications, and may want to consider prescribing routine use of low doses of aspirin if the gastrointestinal effects are tolerable.[27]
Blood
Anemia is by far the most common abnormality of the blood cells which can be caused by a variety of mechanisms. The chronic inflammation caused by RA leads to raised hepcidin levels, leading to anemia of chronic disease where iron is poorly absorbed and also sequestered into macrophages. The red cells are of normal size and color (normocytic and normochromic). A low white blood cell count usually only occurs in people with Felty's syndrome with an enlarged liver and spleen. The mechanism of neutropenia is complex. An increased platelet count occurs when inflammation is uncontrolled.[citation needed]
Other
- Kidneys
Renal amyloidosis can occur as a consequence of untreated chronic inflammation.[28] Treatment with penicillamine and gold salts are recognized causes of membranous nephropathy.[citation needed]
- Eyes
- The eye can be directly affected in the form of episcleritis or scleritis, which when severe can very rarely progress to perforating scleromalacia. Rather more common is the indirect effect of keratoconjunctivitis sicca, which is a dryness of eyes and mouth caused by lymphocyte infiltration of lacrimal and salivary glands. When severe, dryness of the cornea can lead to keratitis and loss of vision. Preventive treatment of severe dryness with measures such as nasolacrimal duct blockage is important.[citation needed]
- Liver
- Liver problems in people with rheumatoid arthritis may be due to the underlying disease process or as a result of the medications used to treat the disease.[29] A coexisting autoimmune liver disease, such as primary biliary cirrhosis or autoimmune hepatitis may also cause problems.[29]
- Neurological
Peripheral neuropathy and mononeuritis multiplex may occur. The most common problem is carpal tunnel syndrome caused by compression of the median nerve by swelling around the wrist. Rheumatoid disease of the spine can lead to myelopathy. Atlanto-axial subluxation can occur, owing to erosion of the odontoid process and/or transverse ligaments in the cervical spine's connection to the skull. Such an erosion (>3mm) can give rise to vertebrae slipping over one another and compressing the spinal cord. Clumsiness is initially experienced, but without due care, this can progress to quadriplegia or even death.[30]
- Constitutional symptoms
Constitutional symptoms including fatigue, low grade fever, malaise, morning stiffness, loss of appetite and loss of weight are common systemic manifestations seen in people with active RA.
- Bones
- Local osteoporosis occurs in RA around inflamed joints. It is postulated to be partially caused by inflammatory cytokines. More general osteoporosis is probably contributed to by immobility, systemic cytokine effects, local cytokine release in bone marrow and corticosteroid therapy.[citation needed]
- Cancer
- The incidence of lymphoma is increased, although it is uncommon and associated with the chronic inflammation, not the treatment of RA.[31][32]
Risk factors
RA is a systemic (whole body) autoimmune disease. Some genetic and environmental factors affect the risk for RA.
Genetic
A family history of RA increases the risk around three to five times; as of 2016 it was estimated that genetics may account for between 40 and 65% of cases of seropositive RA, but only around 20% for seronegative RA.[3] RA is strongly associated with genes of the inherited tissue type major histocompatibility complex (MHC) antigen HLA-DR4 is the major genetic factor implicated – the relative importance varies across ethnic groups.[33]Genome-wide association studies examining single-nucleotide polymorphisms have found around one hundred genes associated with RA risk, with most of them involving the HLA system (particularly HLA-DRB1) which controls recognition of self versus nonself molecules; other mutations affecting co-stimulatory immune pathways, for example CD28 and CD40), cytokine signaling, lymphocyte receptor activation threshold (e.g., PTPN22), and innate immune activation appear to have less influence than HLA mutations.[3]
Environmental
There are established epigenetic and environmental risk factors for RA.[34][3]Smoking is an established risk factor for RA in Caucasian populations, increasing the risk three times compared to non-smokers, particularly in men, heavy smokers, and those who are rheumatoid factor positive.[35] Modest alcohol consumption may be protective.[36]
Silica exposure has been linked to RA.[37]
Negative findings
No infectious agent has been consistently linked with RA and there is no evidence of disease clustering to indicate its infectious cause,[33] but periodontal disease has been consistently associated with RA.[3]
The many negative findings suggest that either the trigger varies, or that it might, in fact, be a chance event inherent with the immune response.[38]
Pathophysiology
RA primarily starts as a state of persistent cellular activation leading to autoimmunity and immune complexes in joints and other organs where it manifests.[citation needed] The initial site of disease is the synovial membrane, where swelling and congestion lead to infiltration by immune cells. Three phases of progression of RA are an initiation phase (due to non-specific inflammation), an amplification phase (due to T cell activation), and chronic inflammatory phase, with tissue injury resulting from the cytokines, IL–1, TNF-alpha and IL–6.[18]
Non-specific inflammation
Factors allowing an abnormal immune response, once initiated, become permanent and chronic. These factors are genetic disorders which change regulation of the adaptive immune response.[3] Genetic factors interact with environmental risk factors for RA, with cigarette smoking as the most clearly defined risk factor.[35][39]
Other environmental and hormonal factors may explain higher risks for women, including onset after childbirth and hormonal medications. A possibility for increased susceptibility is that negative feedback mechanisms – which normally maintain tolerance – are overtaken by positive feedback mechanisms for certain antigens, such as IgG Fc bound by rheumatoid factor and citrullinated fibrinogen bound by antibodies to citrullinated peptides (ACPA - Anti–citrullinated protein antibody). A debate on the relative roles of B-cell produced immune complexes and T cell products in inflammation in RA has continued for 30 years, but neither cell is necessary at the site of inflammation, only autoantibodies to IgGFc, known as rheumatoid factors and ACPA, with ACPA having a 80% specificity for diagnosing RA.[40] As with other autoimmune diseases, people with RA have abnormally glycosylated antibodies, which are believed to promote joint inflammation.[41][page needed]
Amplification in the synovium
Once the generalized abnormal immune response has become established – which may take several years before any symptoms occur – plasma cells derived from B lymphocytes produce rheumatoid factors and ACPA of the IgG and IgM classes in large quantities. These activate macrophages through Fc receptor and complement binding, which is part of the intense inflammation in RA.[42][year needed][page needed] Binding of an autoreactive antibody to the Fc receptors is mediated through the antibody's N-glycans, which are altered to promote inflammation in people with RA.[41][page needed]
This contributes to local inflammation in a joint, specifically the synovium with edema, vasodilation and entry of activated T-cells, mainly CD4 in microscopically nodular aggregates and CD8 in microscopically diffuse infiltrates.[citation needed] Synovial macrophages and dendritic cells function as antigen-presenting cells by expressing MHC class II molecules, which establishes the immune reaction in the tissue.[citation needed]
Chronic inflammation
@media all and (max-width:720px){.mw-parser-output .tmulti>.thumbinner{width:100%!important;max-width:none!important}.mw-parser-output .tmulti .tsingle{float:none!important;max-width:none!important;width:100%!important;text-align:center}}
The disease progresses by forming granulation tissue at the edges of the synovial lining, pannus with extensive angiogenesis and enzymes causing tissue damage.[43] The synovium thickens, cartilage and underlying bone disintegrate, and the joint deteriorates, with raised calprotectin levels serving as a biomarker of these events.[44]
Cytokines and chemokines attract and accumulate immune cells, i.e. activated T- and B cells, monocytes and macrophages from activated fibroblasts, in the joint space. By signalling through RANKL and RANK, they eventually trigger osteoclast production, which degrades bone tissue.[3][45][page needed]
Tumor necrosis factors (TNF alpha) plays a major role and several theories exist on how TNF release happens in RA. Tumor necrosis factor‐alpha (TNF‐α) is a proinflammatory cytokine that plays a pivotal role in regulating the inflammatory response in rheumatoid arthritis (RA).If TNF release is stimulated by B cell products in the form of RF or ACPA -containing immune complexes, through activation of immunoglobulin Fc receptors, then RA can be seen as a form of Type III hypersensitivity.[46][year needed][47] As of 1999, if TNF release is stimulated by T cell products such as interleukin-17 it might be closer to type IV hypersensitivity although this terminology may be getting somewhat dated and unhelpful.[48]
Although TNF appears to be the dominant chemical mediator other cytokines are involved in inflammation in RA, because blocking TNF does not benefit all persons and all tissues, particularly lung disease and nodules may get worse. Blocking IL-1, IL-15 and IL-6 have beneficial effects and IL-17 may be important.[49]
Diagnosis
Imaging

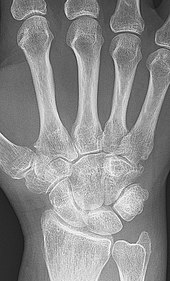
X-ray of the hand in rheumatoid arthritis.
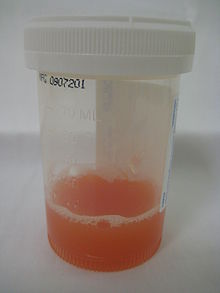
Appearance of synovial fluid from a joint with inflammatory arthritis.
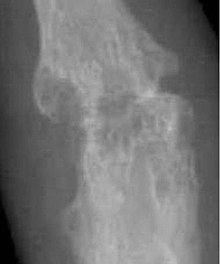
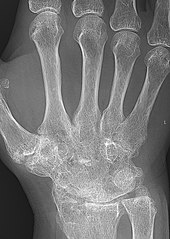
Closeup of bone erosions in rheumatoid arthritis.[50]
X-rays of the hands and feet are generally performed when many joints affected. In RA, there may be no changes in the early stages of the disease or the x-ray may show osteopenia near the joint, soft tissue swelling, and a smaller than normal joint space. As the disease advances, there may be bony erosions and subluxation. Other medical imaging techniques such as magnetic resonance imaging (MRI) and ultrasound are also used in RA.[18][51]
Technical advances in ultrasonography like high-frequency transducers (10 MHz or higher) have improved the spatial resolution of ultrasound images depicting 20% more erosions than conventional radiography. Color Doppler and power Doppler ultrasound are useful in assessing the degree of synovial inflammation as they can show vascular signals of active synovitis. This is important, since in the early stages of RA, the synovium is primarily affected, and synovitis seems to be the best predictive marker of future joint damage.[52]
Blood tests
When RA is clinically suspected, a physician may test for rheumatoid factor (RF) and anti-citrullinated protein antibodies (ACPAs measured as anti-CCP antibodies).[53][page needed]
It is positive in 75-85%, but a negative RF or CCP antibody does not rule out RA, rather, the arthritis is called seronegative, which is in about 15-25% of people with RA.[54] During the first year of illness, rheumatoid factor is more likely to be negative with some individuals becoming seropositive over time. RF is a non-specific antibody and seen in about 10% of healthy people, in many other chronic infections like hepatitis C, and chronic autoimmune diseases such as Sjögren's syndrome and systemic lupus erythematosus. Therefore, the test is not specific for RA.[18]
Hence, new serological tests check for anti-citrullinated protein antibodies ACPAs . These tests are again positive in 61-75% of all RA cases, but with a specificity of around 95%.[55] As with RF, ACPAs are many times present before symptoms have started.[18]
The by far most common clinical test for ACPAs is the anti-cyclic citrullinated peptide (anti CCP) ELISA. In 2008 a serological point-of-care test for the early detection of RA combined the detection of RF and anti-MCV with a sensitivity of 72% and specificity of 99.7%.[56][better source needed][57]
Other blood tests are usually done to differentiate from other causes of arthritis, like the erythrocyte sedimentation rate (ESR), C-reactive protein, full blood count, kidney function, liver enzymes and other immunological tests (e.g., antinuclear antibody/ANA) are all performed at this stage. Elevated ferritin levels can reveal hemochromatosis, a mimic of RA, or be a sign of Still's disease, a seronegative, usually juvenile, variant of rheumatoid arthritis.[citation needed]
Classification Criteria
In 2010 the 2010 ACR / EULAR Rheumatoid Arthritis Classification Criteria were introduced.[58]
The new criterion is not a diagnostic criterion but a classification criterion to identify disease with a high likelihood of developing a chronic form.[18] However a score of 6 or greater unequivocally classifies a person with a diagnosis of rheumatoid arthritis.
These new classification criteria overruled the "old" ACR criteria of 1987 and are adapted for early RA diagnosis. The "new" classification criteria, jointly published by the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) establish a point value between 0 and 10. Four areas are covered in the diagnosis:[58]
- joint involvement, designating the metacarpophalangeal joints, proximal interphalangeal joints, the interphalangeal joint of the thumb, second through fifth metatarsophalangeal joint and wrist as small joints, and shoulders, elbows, hip joints, knees, and ankles as large joints:
- Involvement of 1 large joint gives 0 points
- Involvement of 2–10 large joints gives 1 point
- Involvement of 1–3 small joints (with or without involvement of large joints) gives 2 points
- Involvement of 4–10 small joints (with or without involvement of large joints) gives 3 points
- Involvement of more than 10 joints (with involvement of at least 1 small joint) gives 5 points
- serological parameters – including the rheumatoid factor as well as ACPA – "ACPA" stands for "anti-citrullinated protein antibody":
- Negative RF and negative ACPA gives 0 points
- Low-positive RF or low-positive ACPA gives 2 points
- High-positive RF or high-positive ACPA gives 3 points
- acute phase reactants: 1 point for elevated erythrocyte sedimentation rate, ESR, or elevated CRP value (c-reactive protein)
- duration of arthritis: 1 point for symptoms lasting six weeks or longer
The new criteria accommodate to the growing understanding of RA and the improvements in diagnosing RA and disease treatment. In the "new" criteria serology and autoimmune diagnostics carries major weight, as ACPA detection is appropriate to diagnose the disease in an early state, before joints destructions occur. Destruction of the joints viewed in radiological images was a significant point of the ACR criteria from 1987.[59] This criterion no longer is regarded to be relevant, as this is just the type of damage that treatment is meant to avoid.
In clinical practice, the following criteria apply:[citation needed]
- two or more swollen joints
- morning stiffness lasting more than one hour for at least six weeks
- the detection of rheumatoid factors or autoantibodies against ACPA such as autoantibodies to mutated citrullinated vimentin can confirm the suspicion of RA. A negative autoantibody result does not exclude a diagnosis of RA.
Differential diagnoses
Several other medical conditions can resemble RA, and need to be distinguished from it at the time of diagnosis:[60][61]
- Crystal induced arthritis (gout, and pseudogout) – usually involves particular joints (knee, MTP1, heels) and can be distinguished with an aspiration of joint fluid if in doubt. Redness, asymmetric distribution of affected joints, pain occurs at night and the starting pain is less than an hour with gout.
Osteoarthritis – distinguished with X-rays of the affected joints and blood tests, older age, starting pain less than an hour, asymmetric distribution of affected joints and pain worsens when using joint for longer periods.
Systemic lupus erythematosus (SLE) – distinguished by specific clinical symptoms and blood tests (antibodies against double-stranded DNA)- One of the several types of psoriatic arthritis resembles RA – nail changes and skin symptoms distinguish between them
Lyme disease causes erosive arthritis and may closely resemble RA – it may be distinguished by blood test in endemic areas
Reactive arthritis (previously Reiter's disease) – asymmetrically involves heel, sacroiliac joints and large joints of the leg. It is usually associated with urethritis, conjunctivitis, iritis, painless buccal ulcers, and keratoderma blennorrhagica.
Axial spondyloarthritis (including ankylosing spondylitis) – this involves the spine, although an RA-like symmetrical small-joint polyarthritis may occur in the context of this condition.
Hepatitis C – RA-like symmetrical small-joint polyarthritis may occur in the context of this condition. Hepatitis C may also induce Rheumatoid Factor auto-antibodies
Rarer causes which usually behave differently but may cause joint pains:[60]
Sarcoidosis, amyloidosis, and Whipple's disease can also resemble RA.
Hemochromatosis may cause hand joint arthritis.- Acute rheumatic fever can be differentiated by a migratory pattern of joint involvement and evidence of antecedent streptococcal infection.
- Bacterial arthritis (such as by Streptococcus) is usually asymmetric, while RA usually involves both sides of the body symmetrically.
Gonococcal arthritis (a bacterial arthritis) is also initially migratory and can involve tendons around the wrists and ankles.
Sometimes arthritis is in an undifferentiated stage (i.e. none of the above criteria is positive), even if synovitis is witnessed and assessed with ultrasound imaging.
Monitoring progression
Many tools can be used to monitor remission in rheumatoid arthritis.
- DAS28
Disease Activity Score of 28 joints (DAS28) is widely used as an indicator of RA disease activity and response to treatment. Joints included are (bilaterally): proximal interphalangeal joints (10 joints), metacarpophalangeal joints (10), wrists (2), elbows (2), shoulders (2) and knees (2). When looking at these joints, both the number of joints with tenderness upon touching (TEN28) and swelling (SW28) are counted. The erythrocyte sedimentation rate (ESR) is measured and the affected person makes a subjective assessment (SA) of disease activity during the preceding 7 days on a scale between 0 and 100, where 0 is "no activity" and 100 is "highest activity possible". With these parameters, DAS28 is calculated as:[62]
DAS28=0.56×TEN28+0.28×SW28+0.70×ln(ESR)+0.014×SA{displaystyle DAS28=0.56times {sqrt {TEN28}}+0.28times {sqrt {SW28}}+0.70times ln(ESR)+0.014times SA}
From this, the disease activity of the affected person can be classified as follows:[62]
| Current DAS28 | DAS28 decrease from initial value | |||
|---|---|---|---|---|
> 1.2 | > 0.6 but ≤ 1.2 | ≤ 0.6 | ||
≤ 3.2 | Inactive | Good improvement | Moderate improvement | No improvement |
> 3.2 but ≤ 5.1 | Moderate | Moderate improvement | Moderate improvement | No improvement |
| > 5.1 | Very active | Moderate improvement | No improvement | No improvement |
It is not always a reliable indicator of treatment effect.[63] One major limitation is that low-grade synovitis may be missed.[64]
- Other
- Other tools to monitor remission in rheumatoid arthritis are: ACR-EULAR Provisional Definition of Remission of Rheumatoid arthritis, Simplified Disease Activity Index (SDAI) and Clinical Disease Activity Index (CDAI).[65] Some scores do not require input from a healthcare professional and allow self-monitoring by the person, like HAQ-DI.[66][page needed]
Prevention
There is no known prevention for the condition other than the reduction of risk factors.[67]
Management
There is no cure for RA, but treatments can improve symptoms and slow the progress of the disease. Disease-modifying treatment has the best results when it is started early and aggressively.[68]
The goals of treatment are to minimize symptoms such as pain and swelling, to prevent bone deformity (for example, bone erosions visible in X-rays), and to maintain day-to-day functioning.[69] This is primarily addressed with disease-modifying antirheumatic drugs (DMARDs); analgesics may be used to help manage pain.[5] RA should generally be treated with at least one specific anti-rheumatic medication.[6] The use of benzodiazepines (such as diazepam) to treat the pain is not recommended as it does not appear to help and is associated with risks.[70]
Lifestyle
Regular exercise is recommended as both safe and useful to maintain muscles strength and overall physical function.[71] It is uncertain if specific dietary measures have an effect.[72] Physical activity is beneficial for persons with rheumatoid arthritis complaining of fatigue.[73] Occupational therapy has a positive role to play in improving functional ability of persons with rheumatoid arthritis.[74]
Disease modifying agents
Disease-modifying antirheumatic drugs (DMARDs) are the primary treatment for RA.[6] They are a diverse collection of drugs, grouped by use and convention. They have been found to improve symptoms, decrease joint damage, and improve overall functional abilities.[6] DMARDs should be started early in the disease as they result in disease remission in approximately half of people and improved outcomes overall.[6]
The following drugs are considered as DMARDs: methotrexate, hydroxychloroquine, sulfasalazine, leflunomide, TNF-alpha inhibitors (certolizumab, infliximab and etanercept), abatacept, and anakinra. Rituximab and tocilizumab are monoclonal antibodies and are also DMARDs.[6]
Hydroxychloroquine, apart from its low toxicity profile, is considered effective in the moderate RA treatment.[75]
The most commonly used agent is methotrexate with other frequently used agents including sulfasalazine and leflunomide.[6] Leflunomide is effective when used from 6–12 months, with similar effectivness to methorexate when used for 2 years.[76]Sodium aurothiomalate (gold) and cyclosporin are less commonly used due to more common adverse effects.[6] However, cyclosporin was found to be effective in the progressive RA when used up to one year.[77] Agents may be used in combinations.[6] Methotrexate is the most important and useful DMARD and is usually the first treatment.[6][5][78] Adverse effects should be monitored regularly with toxicity including gastrointestinal, hematologic, pulmonary, and hepatic.[78] Side effects such as nausea, vomiting or abdominal pain can be reduced by taking folic acid.[79]
A 2015 Cochrane review found rituximab with methotrexate to be effective in improving symptoms compared to methotrexate alone.[80] Rituximab works by depicting levels of B-cells (immune cell that is involved in inflammation). People taking rituximab had improved pain, function, reduced disease activity and reduced joint damage based on x-ray images. After 6 months, 21% more people had improvement in their symptoms using rituximab and methotrexate.[80]
Biological agents should generally only be used if methotrexate and other conventional agents are not effective after a trial of three months.[6] They are associated with a higher rate of serious infections as compared to other DMARDs.[81] Biological DMARD agents used to treat rheumatoid arthritis include: tumor necrosis factor alpha (TNFα) blockers such as infliximab; interleukin 1 blockers such as anakinra, monoclonal antibodies against B cells such as rituximab, and tocilizumab T cell co-stimulation blocker such as abatacept. They are often used in combination with either methotrexate or leflunomide.[6][3] Abatacept should not be used at the same time as other biologics.[82] In those who are well controlled on TNF blockers decreasing the dose does not appear to affect overall function.[83] Persons should be screened for latent tuberculosis before starting any TNF blockers therapy to avoid reactivation.[18]
TNF blockers and methotrexate appear to have similar effectiveness when used alone and better results are obtained when used together. Golimumab showed significant effectivness when used with methotraxate.[84] TNF blockers appear to have equivalent effectiveness with etanercept appearing to be the safest.[85] Abatacept appears effective for RA with 20% more people improving with treatment than without but long term safety studies are yet unavailable.[86]Adalimumab slows the time for the radiographic progression when used for 52 weeks.[87] However, there is a lack of evidence to distinguish between the biologics available for RA.[88] Issues with the biologics include their high cost and association with infections including tuberculosis.[3]
Anti-inflammatory and analgesic agents
Glucocorticoids can be used in the short term and at the lowest dose possible for flare-ups and while waiting for slow-onset drugs to take effect.[6][3]
Non-NSAID drugs to relieve pain, like paracetamol may be used to help relieve the pain symptoms; they do not change the underlying disease.[5]
NSAIDs reduce both pain and stiffness in those with RA but do not affect the underlying disease and appear to have no effect on people's long term disease course and thus are no longer first line agents.[3][89] NSAIDs should be used with caution in those with gastrointestinal, cardiovascular, or kidney problems.[90][91][92] Use of methotrexate together with NSAIDS is safe, if adequate monitoring is done.[93]COX-2 inhibitors, such as celecoxib, and NSAIDs are equally effective.[94][95]
The neuromodulator agents topical capsaicin may be reasonable to use in an attempt to reduce pain.[96]Nefopam by mouth and cannabis are not recommended as of 2012 as the risks of use appear to be greater than the benefits.[96]
Surgery
Especially for affected fingers, hands, and wrists, synovectomy may be needed to prevent pain or tendon rupture when drug treatment has failed. Severely affected joints may require joint replacement surgery, such as knee replacement. Postoperatively, physiotherapy is always necessary.[16]:1080, 1103
Alternative medicine
In general, there is not enough evidence to support any complementary health approaches for RA, with safety concerns for some of them. Some mind and body practices and dietary supplements may help people with symptoms and therefore may be beneficial additions to conventional treatments, but there is not enough evidence to draw conclusions.[9] A systematic review of CAM modalities (excluding fish oil) found that " The available evidence does not support their current use in the management of RA.".[97] Studies showing beneficial effects in RA on a wide variety of CAM modalities are often affected by publication bias and are generally not high quality evidence such as randomized controlled trials (RCTs).[8]
A 2005 Cochrane review states that low level laser therapy can be tried to improve pain and morning stiffness due to rheumatoid arthritis as there are few side-effects.[98]
There is some evidence that Tai Chi improves the range of motion of a joint in persons with rheumatoid arthritis.[99] The evidence for acupuncture is inconclusive[100] with it appearing to be equivalent to sham acupuncture.[101]
A Cochrane review in 2002 showed some benefits of the electrical stimulation as a rehabilitation intervention to improve the power of the hand grip and help to resist fatigue.[102]
Dietary supplements
- Fatty acids
Gamma-linolenic acid, an omega-6 fatty acid, may reduce pain, tender joint count and stiffness, and is generally safe.[103] For omega-3 polyunsaturated fatty acids (found in fish oil), a meta-analysis reported a favorable effect on pain, although confidence in the effect was considered moderate. The same review reported less inflammation but no difference in joint function.[104] A review examined the effect of marine oil omega-3 fatty acids on pro-inflammatory eicosanoid concentrations; leukotriene4 (LTB4) was lowered in people with rheumatoid arthritis but not in those with non-autoimmune chronic diseases. (LTB4) increases vascular permeabiltity and stimulates other inflammatory substances.[105] A third meta-analysis looked at fish consumption. The result was a weak, non-statistically significant inverse association between fish consumption and RA.[106] A fourth review limited inclusion to trials in which people eat ≥2.7 g/day for more than three months. Use of pain relief medication was decreased, but improvements in tender or swollen joints, morning stiffness and physical function were not changed.[107] Collectively, the current evidence is not strong enough to determine that supplementation with omega-3 fatty acids or regular consumption of fish are effective treatments for rheumatoid arthritis.[104][105][106][107]
- Herbal
- The American College of Rheumatology states that no herbal medicines have health claims supported by high-quality evidence and thus they do not recommend their use.[108] There is no scientific basis to suggest that herbal supplements advertised as "natural" are safer for use than conventional medications as both are chemicals. Herbal medications, although labelled "natural", may be toxic or fatal if consumed.[108]
Due to the false belief that herbal supplements are always safe, there is sometimes a hesitancy to report their use which may increase the risk of adverse reaction.[8]
The following are under investigation for treatments for RA, based on preliminary promising results (not recommended for clinical use yet): boswellic acid,[109]curcumin,[110]devil's claw,[111][112]Euonymus alatus,[113] and thunder god vine (Tripterygium wilfordii).[114]NCCIH has noted that, "In particular, the herb thunder god vine (Tripterygium wilfordii) can have serious side effects."[9]
There is conflicting evidence on the role of erythropoiesis-stimulating agents for treatment of anemia in persons with rheumatoid arthritis.[115]
Pregnancy
More than 75% of women with rheumatoid arthritis have symptoms improve during pregnancy but might have symptoms worsen after delivery.[18]Methotrexate and leflunomide are teratogenic (harmful to foetus) and not used in pregnancy. It is recommended women of childbearing age should use contraceptives to avoid pregnancy and to discontinue its use if pregnancy is planned.[69][78] Low dose of prednisolone, hydroxychloroquine and sulfasalazine are considered safe in pregnant persons with rheumatoid arthritis.
Vaccinations
People with RA have an increased risk of infections and mortality and recommended vaccinations can reduce these risks.[116] The inactivated influenza vaccine should be received annually.[117] The pneumococcal vaccine should be administered twice for people under the age 65 and once for those over 65.[118] Lastly, the live-attenuated zoster vaccine should be administered once after the age 60, but is not recommended in people on a tumor necrosis factor alpha blocker.[119]
Prognosis
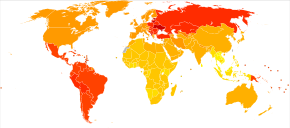
Disability-adjusted life year for RA per 100,000 inhabitants in 2004.[120]
no data
<40
40–50
50–60
60–70
70–80
80–90
90–100
100–110
110–120
120–130
130–140
>140
The course of the disease varies greatly. Some people have mild short-term symptoms, but in most the disease is progressive for life. Around 20%–30% will have subcutaneous nodules (known as rheumatoid nodules); this is associated with a poor prognosis.[citation needed]
Prognostic factors
Poor prognostic factors include,
- Persistent synovitis
- Early erosive disease
- Extra-articular findings (including subcutaneous rheumatoid nodules)
- Positive serum RF findings
- Positive serum anti-CCP autoantibodies
- Carriership of HLA-DR4 "Shared Epitope" alleles
- Family history of RA
- Poor functional status
- Socioeconomic factors
- Elevated acute phase response (erythrocyte sedimentation rate [ESR], C-reactive protein [CRP])
- Increased clinical severity.
Mortality
RA reduces lifespan on average from three to twelve years.[69] Young age at onset, long disease duration, the presence of other health problems, and characteristics of severe RA—such as poor functional ability or overall health status, a lot of joint damage on x-rays, the need for hospitalisation or involvement of organs other than the joints—have been shown to associate with higher mortality.[121] Positive responses to treatment may indicate a better prognosis. A 2005 study by the Mayo Clinic noted that RA sufferers suffer a doubled risk of heart disease,[122] independent of other risk factors such as diabetes, alcohol abuse, and elevated cholesterol, blood pressure and body mass index. The mechanism by which RA causes this increased risk remains unknown; the presence of chronic inflammation has been proposed as a contributing factor.[123] It is possible that the use of new biologic drug therapies extend the lifespan of people with RA and reduce the risk and progression of atherosclerosis.[124] This is based on cohort and registry studies, and still remains hypothetical. It is still uncertain whether biologics improve vascular function in RA or not. There was an increase in total cholesterol and HDLc levels and no improvement of the atherogenic index.[125]
Epidemiology

Deaths from rheumatoid arthritis per million persons in 2012 .mw-parser-output .refbegin{font-size:90%;margin-bottom:0.5em}.mw-parser-output .refbegin-hanging-indents>ul{list-style-type:none;margin-left:0}.mw-parser-output .refbegin-hanging-indents>ul>li,.mw-parser-output .refbegin-hanging-indents>dl>dd{margin-left:0;padding-left:3.2em;text-indent:-3.2em;list-style:none}.mw-parser-output .refbegin-100{font-size:100%}
0–0
1–1
2–3
4–5
6–6
7–8
9–9
10–12
13–20
21–55
RA affects between 0.5 and 1% of adults in the developed world with between 5 and 50 per 100,000 people newly developing the condition each year.[3] In 2010 it resulted in about 49,000 deaths globally.[126]
Onset is uncommon under the age of 15 and from then on the incidence rises with age until the age of 80. Women are affected three to five times as often as men.[18]
The age at which the disease most commonly starts is in women between 40 and 50 years of age, and for men somewhat later.[127] RA is a chronic disease, and although rarely, a spontaneous remission may occur, the natural course is almost invariably one of the persistent symptoms, waxing and waning in intensity, and a progressive deterioration of joint structures leading to deformations and disability.
History
The first known traces of arthritis date back at least as far as 4500 BC. A text dated 123 AD first describes symptoms very similar to RA.[citation needed] It was noted in skeletal remains of Native Americans found in Tennessee.[128] In Europe, the disease is vanishingly rare before the 17th century.[129] The first recognized description of RA in modern medicine was in 1800 by the French physician Dr Augustin Jacob Landré-Beauvais (1772–1840) who was based in the famed Salpêtrière Hospital in Paris.[12] The name "rheumatoid arthritis" itself was coined in 1859 by British rheumatologist Dr Alfred Baring Garrod.[130]
An anomaly has been noticed from the investigation of Pre-Columbian bones. The bones from the Tennessee site show no signs of tuberculosis even though it was prevalent at the time throughout the Americas.[131]
The art of Peter Paul Rubens may possibly depict the effects of RA. In his later paintings, his rendered hands show, in the opinion of some physicians, increasing deformity consistent with the symptoms of the disease.[132][133] RA appears to some to have been depicted in 16th-century paintings.[134] However, it is generally recognized in art historical circles that the painting of hands in the 16th and 17th century followed certain stylized conventions, most clearly seen in the Mannerist movement. It was conventional, for instance, to show the upheld right hand of Christ in what now appears a deformed posture. These conventions are easily misinterpreted as portrayals of disease.
Historic treatments for RA have also included: rest, ice, compression and elevation, apple diet, nutmeg, some light exercise every now and then, nettles, bee venom, copper bracelets, rhubarb diet, extractions of teeth, fasting, honey, vitamins, insulin, magnets, and electroconvulsive therapy (ECT).[135]
Etymology
Rheumatoid arthritis is derived from the Greek word ῥεύμα-rheuma (nom.), ῥεύματος-rheumatos (gen.) ("flow, current"). The suffix -oid ("resembling") gives the translation as joint inflammation that resembles rheumatic fever. Rhuma which means watery discharge might refer to the fact that the joints are swollen or that the disease may be made worse by wet weather.[13]
Research
Meta-analysis found an association between periodontal disease and RA, but the mechanism of this association remains unclear.[136] Two bacterial species associated with periodontitis are implicated as mediators of protein citrullination in the gums of people with RA.[3]
Vitamin D deficiency is more common in people with rheumatoid arthritis than in the general population.[137][138] However, whether vitamin D deficiency is a cause or a consequence of the disease remains unclear.[139] One meta-analysis found that vitamin D levels are low in people with rheumatoid arthritis and that vitamin D status correlates inversely with prevalence of rheumatoid arthritis, suggesting that vitamin D deficiency is associated with susceptibility to rheumatoid arthritis.[140]
References
^ abcdefghijklmnopqrstuvwx "Handout on Health: Rheumatoid Arthritis". National Institute of Arthritis and Musculoskeletal and Skin Diseases. August 2014. Archived from the original on June 30, 2015. Retrieved July 2, 2015..mw-parser-output cite.citation{font-style:inherit}.mw-parser-output q{quotes:"""""""'""'"}.mw-parser-output code.cs1-code{color:inherit;background:inherit;border:inherit;padding:inherit}.mw-parser-output .cs1-lock-free a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/6/65/Lock-green.svg/9px-Lock-green.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-limited a,.mw-parser-output .cs1-lock-registration a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/d/d6/Lock-gray-alt-2.svg/9px-Lock-gray-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-subscription a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/a/aa/Lock-red-alt-2.svg/9px-Lock-red-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration{color:#555}.mw-parser-output .cs1-subscription span,.mw-parser-output .cs1-registration span{border-bottom:1px dotted;cursor:help}.mw-parser-output .cs1-hidden-error{display:none;font-size:100%}.mw-parser-output .cs1-visible-error{font-size:100%}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration,.mw-parser-output .cs1-format{font-size:95%}.mw-parser-output .cs1-kern-left,.mw-parser-output .cs1-kern-wl-left{padding-left:0.2em}.mw-parser-output .cs1-kern-right,.mw-parser-output .cs1-kern-wl-right{padding-right:0.2em}
^ abcdef Majithia V, Geraci SA (2007). "Rheumatoid arthritis: diagnosis and management". Am. J. Med. 120 (11): 936–9. doi:10.1016/j.amjmed.2007.04.005. PMID 17976416.
^ abcdefghijklmn Smolen, Josef S.; Aletaha, Daniel; McInnes, Iain B. (2016-10-22). "Rheumatoid arthritis". Lancet. 388 (10055): 2023–2038. doi:10.1016/S0140-6736(16)30173-8. PMID 27156434.
^ GBD 2015 Mortality and Causes of Death, Collaborators. (8 October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1459–1544. doi:10.1016/S0140-6736(16)31012-1. PMC 5388903. PMID 27733281.
^ abcd "Rheumatoid arthritis in adults: management: recommendations: Guidance and guidelines". NICE. December 2015. Archived from the original on 2017-04-16.
^ abcdefghijklm Singh, JA; Saag, KG; Bridges SL, Jr; Ak l; et al. (January 2016). "2015 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis". Arthritis & Rheumatology. 68 (1): 1–26. doi:10.1002/art.39480. PMID 26545940.CS1 maint: Explicit use of et al. (link)
^ Singh, JA; Wells, GA; Christensen, R; et al. (16 February 2011). "Adverse effects of biologics: a network meta-analysis and Cochrane overview". The Cochrane Database of Systematic Reviews (2): CD008794. doi:10.1002/14651858.CD008794.pub2. PMID 21328309.CS1 maint: Explicit use of et al. (link)
^ abc Efthimiou P, Kukar M (2010). "Complementary and alternative medicine use in rheumatoid arthritis: proposed mechanism of action and efficacy of commonly used modalities". Rheumatology International. 30 (5): 571–86. doi:10.1007/s00296-009-1206-y. PMID 19876631.
^ abc "Rheumatoid Arthritis and Complementary Health Approaches". National Center for Complementary and Integrative Health. January 2006. Archived from the original on July 5, 2015. Retrieved July 1, 2015.
^ GBD 2015 Disease and Injury Incidence and Prevalence, Collaborators. (8 October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
^ GBD 2013 Mortality Causes of Death Collaborators (2015). "Global, regional, and national age–sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013". The Lancet. 385 (9963): 117–171. doi:10.1016/S0140-6736(14)61682-2. PMC 4340604. PMID 25530442.
^ ab Landré-Beauvais AJ (1800). La goutte asthénique primitive (doctoral thesis). Paris. reproduced in Landré-Beauvais AJ (2001). "The first description of rheumatoid arthritis. Unabridged text of the doctoral dissertation presented in 1800". Joint Bone Spine. 68 (2): 130–43. doi:10.1016/S1297-319X(00)00247-5. PMID 11324929.
^ ab Paget, Stephen A.; Lockshin, Michael D.; Loebl, Suzanne (2002). The Hospital for Special Surgery Rheumatoid Arthritis Handbook Everything You Need to Know. New York: John Wiley & Sons. p. 32. ISBN 9780471223344. Archived from the original on 2017-02-22.
^ Turesson C, O'Fallon WM, Crowson CS, Gabriel SE, Matteson EL (2003). "Extra-articular disease manifestations in rheumatoid arthritis: incidence trends and risk factors over 46 years". Ann. Rheum. Dis. 62 (8): 722–7. doi:10.1136/ard.62.8.722. PMC 1754626. PMID 12860726.
^ Cutolo, Maurizio; Kitas, George D.; van Riel, Piet L.C.M. (February 2014). "Burden of disease in treated rheumatoid arthritis patients: Going beyond the joint". Seminars in Arthritis and Rheumatism. 43 (4): 479–488. doi:10.1016/j.semarthrit.2013.08.004. ISSN 0049-0172. PMID 24080116.
^ abcd Walker, Brian R.; Colledge, Nicki R.; Ralston, Stuart H.; Penman, Ian D., eds. (2014). Davidson's principles and practice of medicine (22nd ed.). Churchill Livingstone/Elsevier. ISBN 978-0-7020-5035-0.
^ Gaffo A, Saag KG, Curtis JR (2006). "Treatment of rheumatoid arthritis". Am J Health Syst Pharm. 63 (24): 2451–2465. doi:10.2146/ajhp050514. PMC 5164397. PMID 17158693.
^ abcdefghi Shah, Ankur (2012). Harrison's Principles of Internal Medicine (18th ed.). United States: McGraw Hill. p. 2738. ISBN 978-0-07174889-6.
^ Turesson, C (May 2013). "Extra-articular rheumatoid arthritis". Current Opinion in Rheumatology. 25 (3): 360–6. doi:10.1097/bor.0b013e32835f693f. PMID 23425964.
^ Genta, Marcia S.; Genta, Robert M.; Gabay, Cem (2006-10-01). "Systemic rheumatoid vasculitis: a review". Seminars in Arthritis and Rheumatism. 36 (2): 88–98. doi:10.1016/j.semarthrit.2006.04.006. PMID 17023257.
^ ab Khan Mohammad Beigi, Pooya (2018). Alopecia Areata. doi:10.1007/978-3-319-72134-7. ISBN 978-3-319-72133-0.
^ Kim, Eunice J.; Collard, Harold R.; King, Talmadge E. (2009-11-01). "Rheumatoid arthritis-associated interstitial lung disease: the relevance of histopathologic and radiographic pattern". Chest. 136 (5): 1397–1405. doi:10.1378/chest.09-0444. ISSN 1931-3543. PMC 2818853. PMID 19892679.
^ Balbir-Gurman, Alexandra; Yigla, Mordechai; Nahir, Abraham Menahem; Braun-Moscovici, Yolanda (2006-06-01). "Rheumatoid pleural effusion". Seminars in Arthritis and Rheumatism. 35 (6): 368–378. doi:10.1016/j.semarthrit.2006.03.002. ISSN 0049-0172. PMID 16765714.
^ Wolfe F, Mitchell DM, Sibley JT, Fries JF, Bloch DA, Williams CA, Spitz PW, Haga M, Kleinheksel SM, Cathey MA (April 1994). "The mortality of rheumatoid arthritis". Arthritis Rheum. 37 (4): 481–94. doi:10.1002/art.1780370408. PMID 8147925.
^ Aviña-Zubieta JA, Choi HK, Sadatsafavi M, Etminan M, Esdaile JM, Lacaille D (2008). "Risk of cardiovascular mortality in patients with rheumatoid arthritis: a meta-analysis of observational studies". Arthritis Rheum. 59 (12): 1690–1697. doi:10.1002/art.24092. PMID 19035419.CS1 maint: Multiple names: authors list (link)
^ Alenghat, Francis J. (2016-02-04). "The Prevalence of Atherosclerosis in Those with Inflammatory Connective Tissue Disease by Race, Age, and Traditional Risk Factors". Scientific Reports. 6: 20303. doi:10.1038/srep20303. PMC 4740809. PMID 26842423.
^ ab Gupta A, Fomberstein B (2009). "Evaluating cardiovascular risk in rheumatoid arthritis". Journal of Musculoskeletal Medicine. 26 (8): 481–94. Archived from the original on 2012-07-23.
^ de Groot K (August 2007). "[Renal manifestations in rheumatic diseases]". Internist (Berl). 48 (8): 779–85. doi:10.1007/s00108-007-1887-9. PMID 17571244.
^ ab Selmi, Carlo; Santis, Maria De; Gershwin, M Eric (2011). "Liver involvement in subjects with rheumatic disease". Arthritis Research & Therapy. 13 (3): 226. doi:10.1186/ar3319. PMC 3218873. PMID 21722332.
^ Wasserman, B. R.; Moskovich, R.; Razi, A. E. (2011). "Rheumatoid arthritis of the cervical spine--clinical considerations" (PDF). Bulletin of the Nyu Hospital for Joint Diseases. 69 (2): 136–48. PMID 22035393.
^ Baecklund E, Iliadou A, Askling J, et al. (2006). "Association of chronic inflammation, not its treatment, with increased lymphoma risk in rheumatoid arthritis". Arthritis & Rheumatism. 54 (3): 692–701. doi:10.1002/art.21675. PMID 16508929.
^ Franklin J, Lunt M, Bunn D, Symmons D, Silman A (2006). "Incidence of lymphoma in a large primary care derived cohort of cases of inflammatory polyarthritis". Annals of the Rheumatic Diseases. 65 (5): 617–622. doi:10.1136/ard.2005.044784. PMC 1798140. PMID 16249224.
^ ab Doherty, M; Lanyon, P; Ralston, SH. Musculosketal Disorders-Davidson's Principle of Internal Medicine (20th ed.). Elsevier. pp. 1100–1106.
^ Firestein, Gary S.; McInnes, Iain B. (2017-02-21). "Immunopathogenesis of Rheumatoid Arthritis". Immunity. 46 (2): 183–196. doi:10.1016/j.immuni.2017.02.006. PMC 5385708. PMID 28228278.
(subscription required)
^ ab Sugiyama, D.; Nishimura, K.; Tamaki, K.; Tsuji, G.; Nakazawa, T.; Morinobu, A.; Kumagai, S. (2010). "Impact of smoking as a risk factor for developing rheumatoid arthritis: A meta-analysis of observational studies". Annals of the Rheumatic Diseases. 69 (1): 70–81. doi:10.1136/ard.2008.096487. PMID 19174392.
(subscription required)
^ Liao KP, Alfredsson L, Karlson EW (May 2009). "Environmental influences on risk for rheumatoid arthritis". Current Opinion in Rheumatology. 21 (3): 279–83. doi:10.1097/BOR.0b013e32832a2e16. PMC 2898190. PMID 19318947.
(subscription required)
^ Pollard, Kenneth Michael (11 March 2016). "Silica, Silicosis, and Autoimmunity". Frontiers in Immunology. 7: 97. doi:10.3389/fimmu.2016.00097. PMC 4786551. PMID 27014276.
^ Edwards JC, Cambridge G, Abrahams VM (1999). "Do self-perpetuating B lymphocytes drive human autoimmune disease?". Immunology. 97 (2): 188–96. doi:10.1046/j.1365-2567.1999.00772.x. PMC 2326840. PMID 10447731.
(subscription required)
^ Padyukov L, Silva C, Stolt P, et al. (2004). "A gene-environment interaction between smoking and shared epitope genes in HLA-DR provides a high risk of seropositive rheumatoid arthritis". Arthritis Rheum. 50 (10): 3085–92. doi:10.1002/art.20553. PMID 15476204.
(subscription required)
^ Hua, C; Daien, C. I; Combe, B; Landewe, R (2017). "Diagnosis, prognosis and classification of early arthritis: Results of a systematic review informing the 2016 update of the EULAR recommendations for the management of early arthritis". RMD Open. 3 (1): e000406. doi:10.1136/rmdopen-2016-000406. PMC 5237764. PMID 28155923.
^ ab Maverakis E, Kim K, Shimoda M, Gershwin M, et al. (2015). "Glycans in the immune system and The Altered Glycan Theory of Autoimmunity". J Autoimmun. 57 (6): 1–13. doi:10.1016/j.jaut.2014.12.002. PMC 4340844. PMID 25578468.
(subscription required)
^ Boldt AB, Goeldner I, de Messias-Reason IJ (2012). Relevance of the lectin pathway of complement in rheumatic diseases. Adv Clin Chem. Advances in Clinical Chemistry. 56. pp. 105–53. doi:10.1016/B978-0-12-394317-0.00012-1. ISBN 9780123943170. PMID 22397030.CS1 maint: Multiple names: authors list (link)
(subscription required)
^ Elshabrawy, H. A; Chen, Z; Volin, M. V; Ravella, S; Virupannavar, S; Shahrara, S (2015). "The Pathogenic Role of Angiogenesis in Rheumatoid Arthritis". Angiogenesis. 18 (4): 433–448. doi:10.1007/s10456-015-9477-2. PMC 4879881. PMID 26198292.
^ Abildtrup, M; Kingsley, G. H; Scott, D. L (2015). "Calprotectin as a biomarker for rheumatoid arthritis: A systematic review". The Journal of Rheumatology. 42 (5): 760–70. doi:10.3899/jrheum.140628. PMID 25729036.
^ Chiu, YG; Ritchlin, CT (January 2017). "Denosumab: targeting the RANKL pathway to treat rheumatoid arthritis". Expert Opinion on Biological Therapy. 17 (1): 119–128. doi:10.1080/14712598.2017.1263614. PMC 5794005. PMID 27871200.
(subscription required)
^ "HBP310 Immunology". SUNY Stony Brook Pathology Department. Archived from the original on August 7, 2006. Retrieved September 20, 2008.
^ Ghaffar, Abdul. "Hypersensitivity States". University of South Carolina School of Medicine. Archived from the original on May 18, 2016. Retrieved May 29, 2016.
^ Holmes, N. (1999). "Lecture 14: Hypersensitivity". Immunology Division, Department of Pathology, University of Cambridge. Archived from the original on February 6, 2006. Retrieved September 20, 2008.
^ Gaffen, Sarah L. (2017-01-22). "Role of IL-17 in the Pathogenesis of Rheumatoid Arthritis". Current Rheumatology Reports. 11 (5): 365–370. ISSN 1523-3774. PMC 2811488. PMID 19772832.
(subscription required)
^ Ideguchi, Haruko; Ohno, Shigeru; Hattori, Hideaki; Senuma, Akiko; Ishigatsubo, Yoshiaki (2006). "Bone erosions in rheumatoid arthritis can be repaired through reduction in disease activity with conventional disease-modifying antirheumatic drugs". Arthritis Research & Therapy. 8 (3): R76. doi:10.1186/ar1943. ISSN 1478-6354. PMC 1526642. PMID 16646983.
^ Takase-Minegishi, K; Horita, N; Kobayashi, K; Yoshimi, R; Kirino, Y; Ohno, S; Kaneko, T; Nakajima, H; Wakefield, RJ; Emery, P (1 January 2018). "Diagnostic test accuracy of ultrasound for synovitis in rheumatoid arthritis: systematic review and meta-analysis". Rheumatology (Oxford, England). 57 (1): 49–58. doi:10.1093/rheumatology/kex036. PMID 28340066.
^ Schueller-Weidekamm, Claudia (Apr 29, 2010). "Modern ultrasound methods yield stronger arthritis work-up". Diagnostic Imaging. 32.
^ Westwood OM, Nelson PN, Hay FC (2006). "Rheumatoid factors: what's new?". Rheumatology (Oxford). 45 (4): 379–85. doi:10.1093/rheumatology/kei228. PMID 16418203.
(subscription required)
^ Nishimura K, Sugiyama D, Kogata Y, et al. (2007). "Meta-analysis: diagnostic accuracy of anti-cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis". Ann. Intern. Med. 146 (11): 797–808. doi:10.7326/0003-4819-146-11-200706050-00008. PMID 17548411.
(subscription required)
^ van Venrooij, Walther J.; van Beers, Joyce J. B. C.; Pruijn, Ger J. M. (2011-06-07). "Anti-CCP antibodies: the past, the present and the future". Nature Reviews. Rheumatology. 7 (7): 391–398. doi:10.1038/nrrheum.2011.76. ISSN 1759-4804. PMID 21647203.
(subscription required)
^ Renger F, Bang H, Fredenhagen G, et al. "Anti-MCV Antibody Test for the Diagnosis of Rheumatoid Arthritis Using a POCT-Immunoassay". American College of Rheumatology, 2008 Annual Scientific Meeting, Poster Presentation. Archived from the original on 2010-05-27.
^ Luime, J. J.; Colin, E. M.; Hazes, J M W.; Lubberts, E. (2010). "Does anti-mutated citrullinated vimentin have additional value as a serological marker in the diagnostic and prognostic investigation of patients with rheumatoid arthritis? A systematic review". Annals of the Rheumatic Diseases. 69 (2): 337–344. doi:10.1136/ard.2008.103283. PMID 19289382.
(subscription required)
^ ab Aletaha, D.; Neogi, T.; Silman, A. J.; Funovits, J.; Felson, D. T.; Bingham, C. O.; Birnbaum, N. S.; Burmester, G. R.; Bykerk, V. P.; Cohen, M. D.; Combe, B.; Costenbader, K. H.; Dougados, M.; Emery, P.; Ferraccioli, G.; Hazes, J. M.; Hobbs, K.; Huizinga, T. W.; Kavanaugh, A.; Kay, J.; Kvien, T. K.; Laing, T.; Mease, P.; Menard, H. A.; Moreland, L. W.; Naden, R. L.; Pincus, T.; Smolen, J. S.; Stanislawska-Biernat, E.; et al. (2010). "2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative". Annals of the Rheumatic Diseases. 69 (9): 1580–1588. doi:10.1136/ard.2010.138461. PMID 20699241.
^ Arnett, Frank C.; Edworthy, Steven M.; Bloch, Daniel A.; McShane, Dennis J.; Fries, James F.; Cooper, Norman S.; Healey, Louis A.; Kaplan, Stephen R.; Liang, Matthew H.; Luthra, Harvinder S.; Medsger, Thomas A.; Mitchell, Donald M.; Neustadt, David H.; Pinals, Robert S.; Schaller, Jane G.; Sharp, John T.; Wilder, Ronald L.; Hunder, Gene G. (1988). "The american rheumatism association 1987 revised criteria for the classification of rheumatoid arthritis". Arthritis & Rheumatism. 31 (3): 315–324. doi:10.1002/art.1780310302.
^ ab Berkow R, ed. (1992). The Merck Manual (16th ed.). Merck Publishing Group. pp. 1307–08. ISBN 978-0-911910-16-2.
(subscription required)
^ Lovy MR, Starkebaum G, Uberoi S (1996). "Hepatitis C infection presenting with rheumatic manifestations: a mimic of rheumatoid arthritis". J. Rheumatol. 23 (6): 1238–9. PMID 8782126.
(subscription required)
^ ab Prevoo ML, van 't Hof MA, Kuper HH, van Leeuwen MA, van de Putte LB, van Riel PL (1995). "Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis". Arthritis Rheum. 38 (1): 44–8. doi:10.1002/art.1780380107. PMID 7818570.
(subscription required)
^ Kelly, Janis (22 February 2005) DAS28 not always a reliable indicator of treatment effect in RA Archived 2011-02-25 at the Wayback Machine., Medscape Medical News.
^ Uribea, Liliana; Ceróna, Carmen; Amarilesb, Pedro; Fernando Llanoa, Juan; Restrepob, Margarita; et al. (July–September 2016). "Correlación entre la actividad clínica por DAS-28 y ecografía en pacientes con artritis reumatoide" [Correlation between clinical activity measured by DAS-28 and ultrasound in patients with rheumatoid arthritis]. Revista Colombiana de Reumatología (in Spanish). 23 (3): 159–169. doi:10.1016/j.rcreu.2016.05.002.
^ Yazici, Yusuf (2013). "Tools for monitoring remission in rheumatoid arthritis: any will do, let's just pick one and start measuring". Arthritis Research & Therapy. 15 (1): 104. doi:10.1186/ar4139. PMC 3672754. PMID 23374997.
(subscription required)
^ Bruce, Bonnie; Fries, James F. (2003-01-01). "The Stanford Health Assessment Questionnaire: Dimensions and Practical Applications". Health and Quality of Life Outcomes. 1: 20. doi:10.1186/1477-7525-1-20. ISSN 1477-7525. PMC 165587. PMID 12831398.
(subscription required)
^ Spriggs, Brenda B (2014-09-04). "Rheumatoid Arthritis Prevention". Healthline Networks. Archived from the original on July 27, 2014. Retrieved September 16, 2014.
^ Saag KG, Teng GG, Patkar NM, et al. (2008). "American College of Rheumatology 2008 recommendations for the use of nonbiologic and biologic disease-modifying antirheumatic drugs in rheumatoid arthritis". Arthritis Rheum. 59 (6): 762–84. doi:10.1002/art.23721. PMID 18512708.
^ abc Amy M. Wasserman (2011). "Diagnosis and Management of Rheumatoid Arthritis". American Family Physician. 84 (11): 1245–1252. PMID 22150658.
^ Richards BL, Whittle SL, Buchbinder R (Jan 18, 2012). Richards, Bethan L, ed. "Muscle relaxants for pain management in rheumatoid arthritis". Cochrane Database of Systematic Reviews. 1: CD008922. doi:10.1002/14651858.CD008922.pub2. PMID 22258993.CS1 maint: Multiple names: authors list (link)
^ Hurkmans E, van der Giesen FJ, Vliet Vlieland TP, Schoones J, Van den Ende EC (Oct 7, 2009). Hurkmans, Emalie, ed. "Dynamic exercise programs (aerobic capacity and/or muscle strength training) in patients with rheumatoid arthritis". Cochrane Database of Systematic Reviews (4): CD006853. doi:10.1002/14651858.CD006853.pub2. PMID 19821388.CS1 maint: Multiple names: authors list (link)
^ Hagen KB, Byfuglien MG, Falzon L, Olsen SU, Smedslund G (Jan 21, 2009). Hagen, Kåre Birger, ed. "Dietary interventions for rheumatoid arthritis". Cochrane Database of Systematic Reviews (1): CD006400. doi:10.1002/14651858.CD006400.pub2. PMID 19160281.CS1 maint: Multiple names: authors list (link)
^ Cramp, Fiona (2013). "Non-pharmacological interventions for fatigue in rheumatoid arthritis". Cochrane Database of Systematic Reviews (8): CD008322. doi:10.1002/14651858.CD008322.pub2. PMID 23975674.
^ Steultjens, Esther EMJ (2004). "Occupational therapy for rheumatoid arthritis". Cochrane Database of Systematic Reviews (1): CD003114. doi:10.1002/14651858.CD003114.pub2. hdl:2066/58846. PMID 14974005.
^ Suarez-Almazor, ME; Belseck, E; Shea, B; Homik, J; Wells, G; Tugwell, P (2000). "Antimalarials for treating rheumatoid arthritis". The Cochrane Database of Systematic Reviews (4): CD000959. doi:10.1002/14651858.CD000959. PMID 11034691.
^ Osiri, M; Shea, B; Robinson, V; Suarez-Almazor, M; Strand, V; Tugwell, P; Wells, G (2003). "Leflunomide for treating rheumatoid arthritis". The Cochrane Database of Systematic Reviews (1): CD002047. doi:10.1002/14651858.CD002047. PMID 12535423.
^ Wells, G; Haguenauer, D; Shea, B; Suarez-Almazor, ME; Welch, VA; Tugwell, P (2000). "Cyclosporine for rheumatoid arthritis". The Cochrane Database of Systematic Reviews (2): CD001083. doi:10.1002/14651858.CD001083. PMID 10796412.
^ abc DiPiro, Joseph T., Robert L. Talbert, Gary C. Yee, Gary R. Matzke, Barbara G. Wells, and L. Michael Posey (2008) Pharmacotherapy: a pathophysiologic approach. 7th ed. New York: McGraw-Hill,
ISBN 978-0-07-147899-1.
^ Shea, B.; Swinden, M.V.; Tanjong Ghogomu, E.; Ortiz, Z.; Katchamart, W.; Rader, T.; Bombardier, C.; Wells, George A; Tugwell, Peter (May 31, 2013). "Folic acid and folinic acid for reducing side effects in patients receiving methotrexate for rheumatoid arthritis". The Cochrane Database of Systematic Reviews. 5 (5): CD000951. doi:10.1002/14651858.CD000951.pub2. PMID 23728635.
^ ab Lopez-Olivo, Maria Angeles; Amezaga Urruela, Matxalen; McGahan, Lynda; Pollono, Eduardo N.; Suarez-Almazor, Maria E. (2015-01-20). "Rituximab for rheumatoid arthritis". The Cochrane Database of Systematic Reviews. 1: CD007356. doi:10.1002/14651858.CD007356.pub2. ISSN 1469-493X. PMID 25603545.
^ Singh, Jasvinder A; Cameron, Chris; Noorbaloochi, Shahrzad; Cullis, Tyler; Tucker, Matthew; Christensen, Robin; Ghogomu, Elizabeth Tanjong; Coyle, Doug; Clifford, Tammy; Tugwell, Peter; Wells, George A (May 2015). "Risk of serious infection in biological treatment of patients with rheumatoid arthritis: a systematic review and meta-analysis". The Lancet. 386 (9990): 258–265. doi:10.1016/S0140-6736(14)61704-9. PMC 4580232. PMID 25975452.
^ Maxwell, Lara; Singh, Jasvinder A. (2009-10-07). "Abatacept for rheumatoid arthritis". The Cochrane Database of Systematic Reviews (4): CD007277. doi:10.1002/14651858.CD007277.pub2. ISSN 1469-493X. PMID 19821401.
^ van Herwaarden, N; den Broeder, AA; Jacobs, W; van der Maas, A; Bijlsma, JW; van Vollenhoven, RF; van den Bemt, BH (Sep 29, 2014). "Down-titration and discontinuation strategies of tumor necrosis factor-blocking agents for rheumatoid arthritis in patients with low disease activity". The Cochrane Database of Systematic Reviews. 9 (9): CD010455. doi:10.1002/14651858.CD010455.pub2. PMID 25264908.
^ Singh, JA; Noorbaloochi, S; Singh, G (20 January 2010). "Golimumab for rheumatoid arthritis". The Cochrane Database of Systematic Reviews (1): CD008341. doi:10.1002/14651858.CD008341. PMID 20091667.
^ Aaltonen KJ, Virkki LM, Malmivaara A, Konttinen YT, Nordström DC, Blom M; Virkki; Malmivaara; Konttinen; Nordström; Blom (2012). Hernandez, Adrian V, ed. "Systematic review and meta-analysis of the efficacy and safety of existing TNF blocking agents in treatment of rheumatoid arthritis". PLoS ONE. 7 (1): e30275. doi:10.1371/journal.pone.0030275. PMC 3260264. PMID 22272322.CS1 maint: Multiple names: authors list (link)
^ Maxwell L, Singh JA; Singh (Oct 7, 2009). Maxwell, Lara, ed. "Abatacept for rheumatoid arthritis". Cochrane Database of Systematic Reviews (4): CD007277. doi:10.1002/14651858.CD007277.pub2. PMID 19821401.
^ Navarro-Sarabia, F; Ariza-Ariza, R; Hernandez-Cruz, B; Villanueva, I (20 July 2005). "Adalimumab for treating rheumatoid arthritis". The Cochrane Database of Systematic Reviews (3): CD005113. doi:10.1002/14651858.CD005113.pub2. PMID 16034967.
^ Singh JA, Christensen R, Wells GA, Suarez-Almazor ME, Buchbinder R, Lopez-Olivo MA, Tanjong Ghogomu E, Tugwell P; Christensen; Wells; Suarez-Almazor; Buchbinder; Lopez-Olivo; Tanjong Ghogomu; Tugwell (Oct 7, 2009). Singh, Jasvinder A, ed. "Biologics for rheumatoid arthritis: an overview of Cochrane reviews". Cochrane Database of Systematic Reviews (Submitted manuscript). 128 (4): 309–310. doi:10.1002/14651858.CD007848.pub2. PMID 19821440.CS1 maint: Multiple names: authors list (link)
^ Tarp S, Bartels EM, Bliddal H, Furst DE, Boers M, Danneskiold-Samsøe B, Rasmussen M, Christensen R; Bartels; Bliddal; Furst; Boers; Danneskiold-Samsøe; Rasmussen; Christensen (November 2012). "Effect of nonsteroidal antiinflammatory drugs on the C-reactive protein level in rheumatoid arthritis: a meta-analysis of randomized controlled trials". Arthritis and Rheumatism. 64 (11): 3511–21. doi:10.1002/art.34644. PMID 22833186.CS1 maint: Multiple names: authors list (link)
^ Radner H, Ramiro S, Buchbinder R, Landewé RB, van der Heijde D, Aletaha D; Ramiro; Buchbinder; Landewé; Van Der Heijde; Aletaha (Jan 18, 2012). Radner, Helga, ed. "Pain management for inflammatory arthritis (rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and other spondylarthritis) and gastrointestinal or liver comorbidity". Cochrane Database of Systematic Reviews. 1: CD008951. doi:10.1002/14651858.CD008951.pub2. PMID 22258995.CS1 maint: Multiple names: authors list (link)
^ McCormack PL (2011). "Celecoxib: a review of its use for symptomatic relief in the treatment of osteoarthritis, rheumatoid arthritis and ankylosing spondylitis". Drugs. 71 (18): 2457–89. doi:10.2165/11208240-000000000-00000. PMID 22141388.
^ Marks JL, Colebatch AN, Buchbinder R, Edwards CJ; Colebatch; Buchbinder; Edwards (Oct 5, 2011). Marks, Jonathan L, ed. "Pain management for rheumatoid arthritis and cardiovascular or renal comorbidity". Cochrane Database of Systematic Reviews (10): CD008952. doi:10.1002/14651858.CD008952.pub2. PMID 21975789.CS1 maint: Multiple names: authors list (link)
^ Colebatch, AN; Marks, Jonathan L; Edwards, Christopher J (2011). "Safety of non-steroidal anti-inflammatory drugs, including aspirin and paracetamol (acetaminophen) in people receiving methotrexate for inflammatory arthritis (rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, other spondyloarthritis)". Cochrane Database of Systematic Reviews (11): CD008872. doi:10.1002/14651858.CD008872.pub2. PMID 22071858.
^ Chen YF, Jobanputra P, Barton P, Bryan S, Fry-Smith A, Harris G, Taylor RS; Jobanputra; Barton; Bryan; Fry-Smith; Harris; Taylor (April 2008). "Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and lumiracoxib) for osteoarthritis and rheumatoid arthritis: a systematic review and economic evaluation". Health Technology Assessment (Winchester, England). 12 (11): 1–278, iii. doi:10.3310/hta12110. PMID 18405470.CS1 maint: Multiple names: authors list (link)
^ Fidahic, M; Jelicic Kadic, A; Radic, M; Puljak, L (9 June 2017). "Celecoxib for rheumatoid arthritis". The Cochrane Database of Systematic Reviews. 6: CD012095. doi:10.1002/14651858.CD012095.pub2. PMID 28597983.
^ ab Richards, BL; Whittle, SL; Buchbinder, R (18 January 2012). "Neuromodulators for pain management in rheumatoid arthritis". The Cochrane Database of Systematic Reviews. 1: CD008921. doi:10.1002/14651858.CD008921.pub2. PMID 22258992.
^ Macfarlane GJ, El-Metwally A, De Silva V, Ernst E, Dowds GL, Moots RJ; El-Metwally; De Silva; Ernst; Dowds; Moots; Arthritis Research UK Working Group on Complementary Alternative Medicines (2011). "Evidence for the efficacy of complementary and alternative medicines in the management of rheumatoid arthritis: a systematic review". Rheumatology (Oxford). 50 (9): 1672–83. doi:10.1093/rheumatology/ker119. PMID 21652584.CS1 maint: Multiple names: authors list (link)
^ Brosseau L, Robinson V, Wells G, Debie R, Gam A, Harman K, Morin M, Shea B, Tugwell P (2005). "Low level laser therapy (Classes I, II and III) for treating rheumatoid arthritis". Cochrane Database Syst Rev. 4 (4): CD002049. doi:10.1002/14651858.CD002049.pub2. PMID 16235295.
^ Han, Alcie; Judd, Maria; Welch, Vivian; Wu, Taixiang; Tugwell, Peter; Wells, George A (2004). "Tai chi for treating rheumatoid arthritis". Cochrane Database of Systematic Reviews (3): CD004849. doi:10.1002/14651858.CD004849. PMID 15266544.
^ Lee MS, Shin B-C, Ernst E; Shin; Ernst (2008). "Acupuncture for rheumatoid arthritis: a systematic review". Rheumatology. 47 (12): 1747–53. doi:10.1093/rheumatology/ken330. PMID 18710899.CS1 maint: Multiple names: authors list (link)
^ Macfarlane GJ, Paudyal P, Doherty M, Ernst E, Lewith G, MacPherson H, Sim J, Jones GT; Paudyal; Doherty; Ernst; Lewith; MacPherson; Sim; Jones; Arthritis Research UK Working Group on Complementary Alternative Therapies for the Management of the Rheumatic Diseases (2012). "A systematic review of evidence for the effectiveness of practitioner-based complementary and alternative therapies in the management of rheumatic diseases: rheumatoid arthritis". Rheumatology. 51 (9): 1707–13. doi:10.1093/rheumatology/kes133. PMID 22661556.CS1 maint: Multiple names: authors list (link)
^ Brosseau, LU; Pelland, LU; Casimiro, LY; Robinson, VI; Tugwell, PE; Wells, GE (2002). "Electrical stimulation for the treatment of rheumatoid arthritis". The Cochrane Database of Systematic Reviews (2): CD003687. doi:10.1002/14651858.CD003687. PMID 12076504.
^ Soeken, K L; Miller, S A; Ernst, E (2003). "Herbal medicines for the treatment of rheumatoid arthritis: a systematic review". Centre for Reviews and Dissemination. National Institute for Health Research. Archived from the original on January 16, 2014. Retrieved March 23, 2013.
^ ab Senftleber NK, Nielsen SM, Andersen JR, Bliddal H, Tarp S, Lauritzen L, Furst DE, Suarez-Almazor ME, Lyddiatt A, Christensen R (2017). "Marine Oil Supplements for Arthritis Pain: A Systematic Review and Meta-Analysis of Randomized Trials". Nutrients. 9 (1): 42. doi:10.3390/nu9010042. PMC 5295086. PMID 28067815.
^ ab Jiang J, Li K, Wang F, Yang B, Fu Y, Zheng J, Li D (2016). "Effect of Marine-Derived n-3 Polyunsaturated Fatty Acids on Major Eicosanoids: A Systematic Review and Meta-Analysis from 18 Randomized Controlled Trials". PLoS ONE. 11 (1): e0147351. doi:10.1371/journal.pone.0147351. PMC 4726565. PMID 26808318.
^ ab Di Giuseppe D, Crippa A, Orsini N, Wolk A (2014). "Fish consumption and risk of rheumatoid arthritis: a dose-response meta-analysis". Arthritis Res. Ther. 16 (5): 446. doi:10.1186/s13075-014-0446-8. PMC 4201724. PMID 25267142.
^ ab Lee YH, Bae SC, Song GG (2012). "Omega-3 polyunsaturated fatty acids and the treatment of rheumatoid arthritis: a meta-analysis". Arch. Med. Res. 43 (5): 356–62. doi:10.1016/j.arcmed.2012.06.011. PMID 22835600.
^ ab "Herbal Remedies, Supplements and Acupuncture for Arthritis". American College of Rheumatology. Archived from the original on May 5, 2013. Retrieved May 3, 2013.
^ Abdel-Tawab M, Werz O, Schubert-Zsilavecz M; Werz; Schubert-Zsilavecz (June 2011). "Boswellia serrata: an overall assessment of in vitro, preclinical, pharmacokinetic and clinical data". Clinical Pharmacokinetics. 50 (6): 349–69. doi:10.2165/11586800-000000000-00000. PMID 21553931.CS1 maint: Multiple names: authors list (link)
^ White B, Judkins DZ; Judkins (March 2011). "Clinical Inquiry. Does turmeric relieve inflammatory conditions?". The Journal of Family Practice. 60 (3): 155–6. PMID 21369559.
^ Wegener, T. (1999). "Therapie degenerativer Erkrankungen des Bewegungsapparates mit südafrikanischer Teufelskralle (Harpagophytum procumbens DC)" [Therapy of degenerative diseases of the musculoskeletal system with South African devil's claw (Harpagophytum procumbens DC)]. Wiener Medizinische Wochenschrift (in German). 149 (8–10): 254–257. PMID 10483693.
^ Denner SS (2007). "A review of the efficacy and safety of devil's claw for pain associated with degenerative musculoskeletal diseases, rheumatoid, and osteoarthritis". Holist Nurs Pract. 21 (4): 203–7. doi:10.1097/01.HNP.0000280932.65581.72. PMID 17627199.
^ Zhang LF, Zhao JX; Zhao (December 2005). "The recent research situation of Euonymus alatus". Zhongguo Zhong Yao Za Zhi = Zhongguo Zhongyao Zazhi = China Journal of Chinese Materia Medica. 30 (24): 1895–8. PMID 16494017.
^ Bao J, Dai SM; Dai (2011). "A Chinese herb Tripterygium wilfordii Hook F in the treatment of rheumatoid arthritis: mechanism, efficacy, and safety". Rheumatol. Int. 31 (9): 1123–9. doi:10.1007/s00296-011-1841-y. PMID 21365177.
^ Martí-Carvajal, Arturo J; Agreda-Pérez, Luis H; Solà, Ivan; Simancas-Racines, Daniel (2013). "Erythropoiesis-stimulating agents for anemia in rheumatoid arthritis". Cochrane Database of Systematic Reviews (2): CD000332. doi:10.1002/14651858.CD000332.pub3. PMID 23450527.
^ Perry, Lisa M.; Winthrop, Kevin L.; Curtis, Jeffrey R. (13 June 2014). "Vaccinations for Rheumatoid Arthritis". Current Rheumatology Reports. 16 (8): 431. doi:10.1007/s11926-014-0431-x. PMC 4080407. PMID 24925587.
^ Grohskopf, LA; Olsen, SJ; Sokolow, LZ; Bresee, JS; Cox, NJ; Broder, KR; Karron, RA; Walter, EB; Centers for Disease Control and, Prevention (15 August 2014). "Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP) – United States, 2014–15 influenza season". MMWR. Morbidity and Mortality Weekly Report. 63 (32): 691–7. PMC 4584910. PMID 25121712.
^ Black, CL; Yue, X; Ball, SW; Donahue, SM; Izrael, D; de Perio, MA; Laney, AS; Lindley, MC; Graitcer, SB; Lu, PJ; Williams, WW; Bridges, CB; DiSogra, C; Sokolowski, J; Walker, DK; Greby, SM (19 September 2014). "Influenza vaccination coverage among health care personnel – United States, 2013–14 influenza season". MMWR. Morbidity and Mortality Weekly Report. 63 (37): 805–11. PMC 5779456. PMID 25233281.
^ Hales, CM; Harpaz, R; Ortega-Sanchez, I; Bialek, SR; Centers for Disease Control and Prevention, (CDC) (22 August 2014). "Update on recommendations for use of herpes zoster vaccine". MMWR. Morbidity and Mortality Weekly Report. 63 (33): 729–31. PMC 5779434. PMID 25144544.
^ "WHO Disease and injury country estimates". World Health Organization. 2009. Archived from the original on November 11, 2009. Retrieved November 11, 2009.
^ Kitas, George (4 April 2006) Why is life span shortened by Rheumatoid Arthritis? National Rheumatoid Arthritis Society
^ Rheumatoid Arthritis Patients Have Double the Risk of Heart Failure. mayoclinic.org (3 February 2005).
^ "Cardiac disease in rheumatoid arthritis". Johns Hopkins University. 2002. Archived from the original on October 9, 2006.
^ Atzeni F, Turiel M, Caporali R, Cavagna L, Tomasoni L, Sitia S, Sarzi-Puttini P; Turiel; Caporali; Cavagna; Tomasoni; Sitia; Sarzi-Puttini (2010). "The effect of pharmacological therapy on the cardiovascular system of patients with systemic rheumatic diseases". Autoimmun Rev. 9 (12): 835–9. doi:10.1016/j.autrev.2010.07.018. PMID 20678592.CS1 maint: Multiple names: authors list (link)
^ Damjanov, N; Nurmohamed, MT; Szekanecz, Z (Mar 18, 2014). "Biologics, cardiovascular effects and cancer". BMC Medicine. 12 (1): 48. doi:10.1186/1741-7015-12-48. PMC 3984692. PMID 24642038.
^ Lozano, 1R; Naghavi, M; Foreman, K; Lim, S; Shibuya, K; Aboyans, V; Abraham, J; Adair, T; et al. (Dec 15, 2012). "Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010". Lancet. 380 (9859): 2095–128. doi:10.1016/S0140-6736(12)61728-0. hdl:10536/DRO/DU:30050819. PMID 23245604.
^ Alamanos Y, Voulgari PV, Drosos AA; Voulgari; Drosos (2006). "Incidence and prevalence of rheumatoid arthritis, based on the 1987 American College of Rheumatology criteria: a systematic review". Semin. Arthritis Rheum. 36 (3): 182–8. doi:10.1016/j.semarthrit.2006.08.006. PMID 17045630.CS1 maint: Multiple names: authors list (link)
^ Rothschild, Bruce M. "Tennessee Origins of Rheumatoid Arthritis". Mcclungmuseum.utk.edu. Archived from the original on February 2, 2012. Retrieved March 3, 2011.
^ "Bones of contention". Arthritis Research UK. April 1999. Archived from the original on February 19, 2003. Retrieved February 5, 2013.
^ Garrod AB (1859). The Nature and Treatment of Gout and Rheumatic Gout. London: Walton and Maberly.
^ Rothschild BM, Rothschild C, Helbling M; Rothschild; Helbling (2003). "Unified theory of the origins of erosive arthritis: conditioning as a protective/directing mechanism?". J. Rheumatol. 30 (10): 2095–102. PMID 14528501.CS1 maint: Multiple names: authors list (link)
^ Appelboom T, de Boelpaepe C, Ehrlich GE, Famaey JP; De Boelpaepe; Ehrlich; Famaey (1981). "Rubens and the question of antiquity of rheumatoid arthritis". JAMA. 245 (5): 483–6. doi:10.1001/jama.245.5.483. PMID 7005475.CS1 maint: Multiple names: authors list (link)
^ Kelly, Janis (14 June 2005). "Did RA travel from New World to Old? The Rubens connection". Medscape. Archived from the original on 15 December 2013. Retrieved March 3, 2011.
^ Dequeker J.; Rico H. (1992). "Rheumatoid arthritis-like deformities in an early 16th-century painting of the Flemish-Dutch school". JAMA. 268 (2): 249–251. doi:10.1001/jama.268.2.249. PMID 1608144.
^ Hart FD (1976). "History of the treatment of rheumatoid arthritis". Br Med J. 1 (6012): 763–5. doi:10.1136/bmj.1.6012.763. PMC 1639217. PMID 177148.
^ Tang, Qingqin; Fu, Haitao; Qin, Baodong; Hu, Zhide; Liu, Yaoting; Liang, Yan; Zhou, Lin; Yang, Zaixing; Zhong, Renqian (2017-02-01). "A Possible Link Between Rheumatoid Arthritis and Periodontitis: A Systematic Review and Meta-analysis". The International Journal of Periodontics & Restorative Dentistry. 37 (1): 79–86. doi:10.11607/prd.2656. PMID 27977821.
^ Gatenby P, Lucas R, Swaminathan A (2013). "Vitamin D deficiency and risk for rheumatic diseases: an update". Curr Opin Rheumatol. 25 (2): 184–91. doi:10.1097/BOR.0b013e32835cfc16. PMID 23370372.
^ Wen H, Baker JF (March 2011). "Vitamin D, immunoregulation, and rheumatoid arthritis". Journal of Clinical Rheumatology. 17 (2): 102–7. doi:10.1097/RHU.0b013e31820edd18. PMID 21364350.
^ Guillot X, Semerano L, Saidenberg-Kermanac'h N, Falgarone G, Boissier MC (2010). "Vitamin D and inflammation". Joint Bone Spine. 77 (6): 552–7. doi:10.1016/j.jbspin.2010.09.018. PMID 21067953.
^ Lee, Y. H.; Bae, SC (2016). "Vitamin D level in rheumatoid arthritis and its correlation with the disease activity: A meta-analysis". Clinical and Experimental Rheumatology. 34 (5): 827–833. PMID 27049238. Archived from the original on 2017-04-05.
External links
| Classification | D
|
|---|---|
| External resources |
|
| Wikimedia Commons has media related to Rheumatoid arthritis. |
Rheumatoid arthritis at Curlie
Charles Weber. "History of rheumatoid arthritis".
